Invasion and metastasis are characteristics that determine that a locally-growing cancer is transformed into a systemic process that may endanger the patient’s life. Metastasis in the brain is common in breast cancer and one of the most common causes of death by cancer. To reach the brain, breast cancer cells must migrate from the tumour where they originated, enter the bloodstream, and cross another barrier that protects the brain in a special way, and so most of them die before implanting themselves in the brain.
Each and every one of these steps involves mechanical alterations in the tumour cells. They have to modify their shape and squeeze themselves to move through the confined spaces available, as does an octopus to hide in unlikely places. They also have to release proteins that, like a chemical drill, allow them to undo the barriers that arise along their way. And finally, in the case of breast cancer cells that metastasize in brain, they need proteins, serpins, that cancel the brain’s natural defences, allowing the tumour to grow in its new location.
Researchers of the Laboratory of Molecular Physiology at the Department of Experimental and Health Sciences (CEXS) at UPF, led by Miguel Ángel Valverde, have identified that a low level of the Piezo2 channel hampers the secretion of serpins, invasion and proliferation; while a high level favours them. The research is published this week in the journal Proceedings of the National Academy of Sciences and involves researchers from the Institute for Bioengineering of Catalonia (IBEC) and the universities of Grenoble (France) and Johns Hopkins (USA).

High levels of the Piezo2 channel favour the invasion and survival of metastatic cells
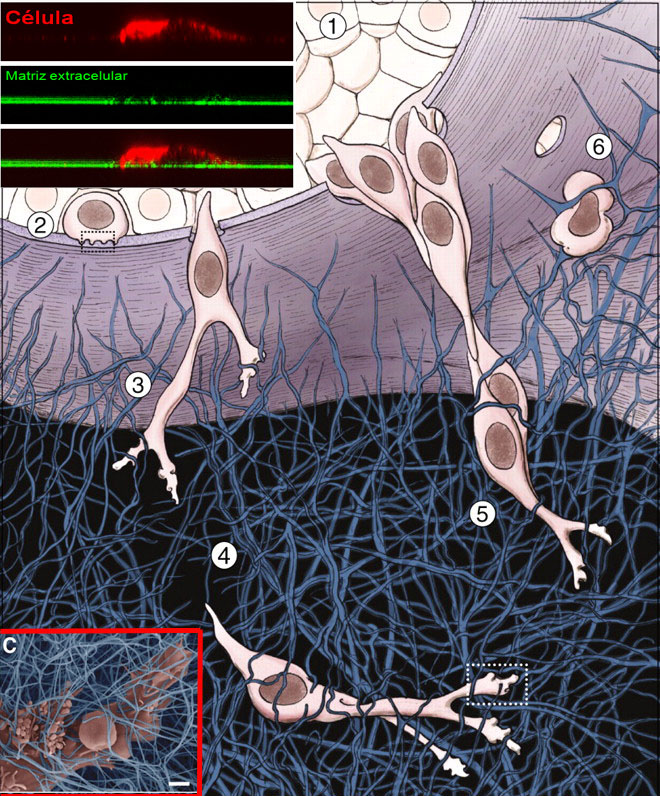
“Our initial goal was to find out what was the reason for breast cancer cells that produce metastasis in the brain to release serpins, so that they override the defences of the brain”, explains Carlos Pardo-Pastor, a trainee researcher of the Laboratory of Molecular Physiology at UPF and first author of the article. “During the journey of a metastatic cell, it must adapt to numerous changes in the physical and mechanical properties of its environment. Therefore, we postulate that the ion channels that detect mechanical and osmotic changes in the cells may be relevant for metastasis”, adds Miguel Ángel Valverde.
The study, carried out with breast cancer cell cultures, analysed the presence of many ion channels which could be used to explore the physical environment of the cells and found that breast cancer cells that specifically metastasize in the brain present higher levels of Piezo2 channel than the cells that have no predilection for this location of metastasis.
Ion channels are selective gates through which the ions enter and leave the cell. In the case of Piezo channels, their opening allows the flow of calcium when the cell detects changes in the rigidity of the environment or when it passes through overly narrow spaces (known as cell confinement). According to the study, this sign of calcium, in turn, triggers a broad range of responses, from the organization of the cell skeleton structure to the activation of factors that control the necessary gene expression as to maintain cell proliferation, the secretion of serpins, and the generation of structures known as invadosomes, necessary to pierce the extracellular matrix and allow cells to pass.
The discovery could turn these channels into the target of new drugs that reduce the risk of metastasis.
This research has been funded, among other sources, by the Ministry of Economy, Industry and Competitiveness, ERDF and the Maria de Maeztu Programme for units of excellence in research and development.
Reference article:
Carlos Pardo-Pastor, Fanny Rubio-Moscardo, Marina Vogel-González, Selma A. Serra, Alexandros Afthinos, Sanela Mrkonjic, Olivier Destaing, Juan F. Abenza, José M. Fernández-Fernández, Xavier Trepat, Corinne Albiges-Rizo, Konstantinos Konstantopoulos, and Miguel A. Valverdea. Piezo2 channel regulates RhoA and actin cytoskeleton to promote cell mechanobiological responses. Proceedings of the National Academy of Sciences U.S.A. February 2018