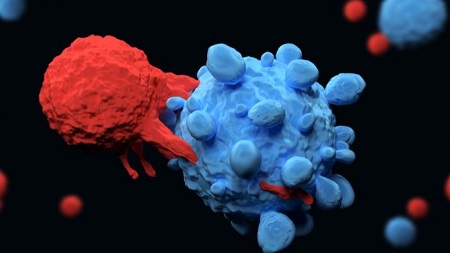
While the success of autologous chimeric antigen receptors (CAR) T-cell medicines cannot be denied, with the recent approvals of Kymriah (tisagenlecleucel) and Yescarta (axicabtagene ciloleucel) paving the way, there are still some limitations to use autologous cells as starting materials including cost, production time as well as possible impacts of the cancer patient’s own cells on the quality and outcomes. For this reason, developing strategies to adapt allogeneic cells for CAR T therapy has several interesting advantages.
Biopharmaceutical company Celyad recently obtained FDA approval for its Investigational New Drug (IND) application for CYAD-101, the first non–gene-edited allogeneic clinical program targeting patients with unresectable colorectal cancer in combination with standard chemotherapy.
More recently, Cellectis received FDA approval for an IND to initiate a Phase 1 clinical trial for UCARTCS1 in patients with multiple myeloma (MM) further widening the possibilities and momentum behind allogeneic CAR T cells.

El gen AtCDF3 promueve una mayor producción de az...

Un estudio con datos de los últimos 35 años, ind...
Un equipo de investigadores de la Universidad Juli...

En nuestro post hablamos sobre este interesante tipo de célula del si...

Palobiofarma S.L. se complace en anunciar la finalización del tratami...
Portal de biotecnología en España
¡Suscríbase a nuestro newsletter para estar al día con las últimas noticias y ofertas!
2013 © Biotech-Spain.com - Site Developments SL. Todos los derechos reservados. Terminos y Condiciones | Política de Privacidad
Articles
Directory