Advances in clinical pharmacology and cancer research are fueling the rise of high potency active pharmaceutical ingredients (HPAPIs).
A significant proportion of new drugs contain HPAPIs, which is leading to a spectacular increase in demand. Manufacturing HPAPIs is a complex process involving clean room processing operations with containment equipment and facilities. These processes are carried out at negative pressure to avoid environmental contamination and workers must also wear full protective equipment. In addition, decontamination systems such as mist or air showers should be used in changing room areas.
At Albian Group we offer our clients the design, manufacture and validation of clean rooms for their HPAPIs projects, as well as any necessary equipment, always focusing on safety. We make sure that the facilities developed comply with the GMP regulations.

The research team observed changes in head circumf...

AtCDF3 gene induced greater production of sugars a...

Un estudio con datos de los últimos 35 años, ind...

En nuestro post hablamos sobre este interesante tipo de célula del...
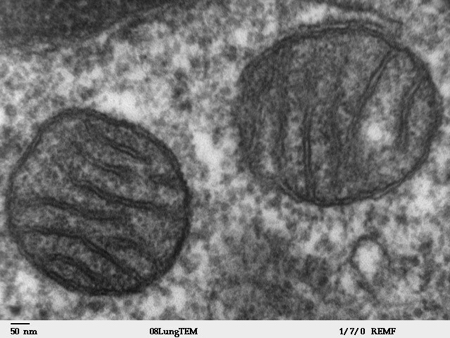
Research led by IIBB-CSIC and CIBEREHD scientists identifies S-adenosy...
Biotechnology portal in Spain
Subscribe to our newsletter and stay up to date with the latest news and deals!
2013 © Biotech-Spain.com - Site Developments SL. All Rights Reserved. Terms of Service | Privacy Policy
Articles
Directory
