On the 3rd of August 2017, the Committee of the Eurasian Economic Union (EAEU) released the standards for EAEU electronic submissions.
The Board of the EAEU has published decisions on the electronic submission of drug applications. These include decisions 78 and 79 of the Board:
What does it mean for the applicant? What is the to-do list?
For all your existing applications already approved in one of the EAEU member states, you should start getting prepared for the transition to the electronic format by:
Additionally, it is advised that you submit a baseline submission of your existing dossier in electronic form.
If you don’t have an existing and approved application within one of the EAEU member states, you are now able to start your application directly in the electronic format.

El equipo de investigadores observó cambios en el...

El gen AtCDF3 promueve una mayor producción de az...

Un estudio con datos de los últimos 35 años, ind...

En nuestro post hablamos sobre este interesante tipo de célula del si...
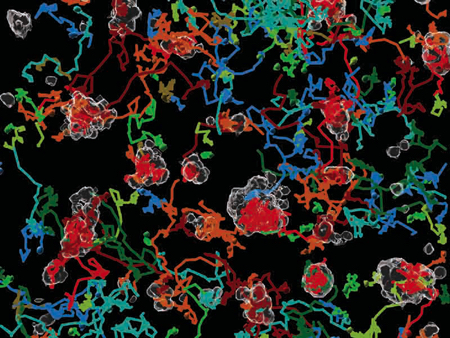
La revista ‘Nature Protocols’ selecciona esta técnica como “pro...
Portal de biotecnología en España
¡Suscríbase a nuestro newsletter para estar al día con las últimas noticias y ofertas!
2013 © Biotech-Spain.com - Site Developments SL. Todos los derechos reservados. Terminos y Condiciones | Política de Privacidad
Articles
Directory
