In September 2019, the European Commission (EC) requested EMA to start a review of ranitidine medicines after tests showed that some of these products contained the impurity N-nitrosodimethylamine (NDMA). Ranitidine medicines are used widely to reduce the production of stomach acid in patients with conditions such as heartburn and stomach ulcers.
According to the ongoing assessment, the rapporteurs of the ranitidine referral consider that the benefit-risk balance of medicinal products containing ranitidine cannot be concluded as being positive and proposed to suspend all marketing authorisations with conditions to lift the suspension.
In the meantime, FDA has requested to all marketing authorisation holders (MAHs) to withdraw the marketing authorisations in US and ranitidine will no longer be available in US as OTC nor Rx prescriptions.
If you need any help, please contact us at info@asphalion.com

El equipo de investigadores observó cambios en el...

El gen AtCDF3 promueve una mayor producción de az...

Un estudio con datos de los últimos 35 años, ind...

En nuestro post hablamos sobre este interesante tipo de célula del si...
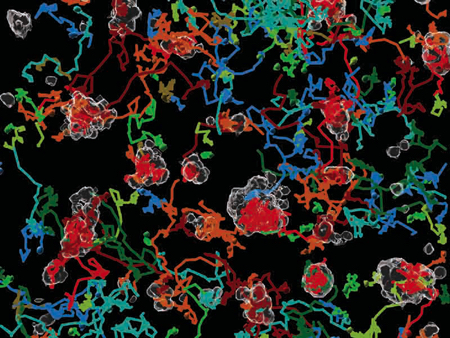
La revista ‘Nature Protocols’ selecciona esta técnica como “pro...
Portal de biotecnología en España
¡Suscríbase a nuestro newsletter para estar al día con las últimas noticias y ofertas!
2013 © Biotech-Spain.com - Site Developments SL. Todos los derechos reservados. Terminos y Condiciones | Política de Privacidad
Articles
Directory
