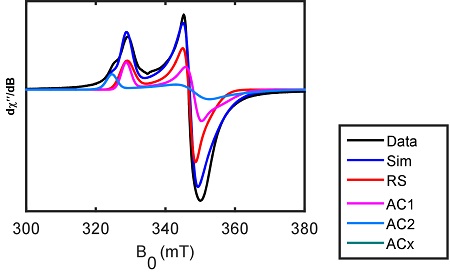
CBGP researchers, in collaboration with the group of David Britt at the University of California-Davis, show that the nitrogenase iron-molybdenum cofactor (FeMo-co) maturase NifB carries three [4Fe-4S] clusters. In addition to the catalytic SAM binding cluster, NifB carries two auxiliary [4Fe-4S] clusters proposed to serve as substrates for the biosynthesis of the Fe8S9C core of FeMo-co.
Spectroscopic evidence indicates coordination of a SAM molecule (acting as methyl donor) to one of the auxiliary clusters, as well as N coordination to at least one of the auxiliary clusters, which permits a mechanism to release the FeS cluster product through protonation.
This analysis represents concise and novel data to understand the mechanism of synthesis of the Fe8S9C core of FeMo-co.
Original Paper:
Wilcoxen, J; Arragain, S; Scandurra, AA; Jimenez-Vicente, E; Echavarri-Erasun, C; Pollmann, S; Britt, RD; Rubio, LM. 2016. "Electron paramagnetic resonance characterization of three iron–sulfur clusters present in the nitrogenase cofactor maturase NifB from Methanocaldococcus infernus". Journal of the American Chemical Society. DOI: 10.1021/jacs.6b03329".

La mejor actitud que podemos adoptar es la de trat...

The research team observed changes in head circumf...

AtCDF3 gene induced greater production of sugars a...

En nuestro post hablamos sobre este interesante tipo de célula del...

Many repetitive regions of the genome have been considered “junk DNA...
Biotechnology portal in Spain
Subscribe to our newsletter and stay up to date with the latest news and deals!
2013 © Biotech-Spain.com - Site Developments SL. All Rights Reserved. Terms of Service | Privacy Policy
Articles
Directory