Oryzon Genomics, S.A. (ISIN Code: ES0167733015, ORY), a clinical-stage biopharmaceutical company leveraging epigenetics to develop therapies in diseases with strong unmet medical need, today reported financial results for the fourth quarter of 2021 and provided an update on recent developments.
Dr Carlos Buesa, Oryzon’s Chief Executive Officer, said: “We made strong progress on our clinical pipeline in 2021. In oncology, we completed the full enrollment of patients in iadademstat’s Phase II trial in acute myeloid leukemia (AML) and presented new positive clinical data from this trial, most recently at ASH in December, supporting the strong clinical activity of iadademstat. Also, we have unfolded our registrational strategy in AML and in Extensive-Disease Small Cell Lung Cancer (ED-SCLC) with two new carefully designed trials to start in 2022, FRIDA and STELLAR, that we believe may merit accelerated approval.”
“In CNS, we also reached important milestones. Vafidemstat’s Phase IIb PORTICO in BPD concluded its deployment phase, we received IND approval from the FDA in June and we are now actively recruiting patients in the USA, Spain, Germany, Bulgaria and Serbia. We have also obtained approval and initiated recruitment for our vafidemstat’s Phase IIb in schizophrenia, called EVOLUTION. Also, together with the most prestigious KOLs in the space, we are progressing the design of HOPE, the first randomized Phase I/II personalized medicine trial with an LSD1 inhibitor, in particular in Kabuki Syndrome patients, which we expect to start in the first half of 2022. Oryzon has further strengthened its permanent presence in the US with the appointment of two new US-based executives: we welcome Sai Nandi as our Global Chief Business Officer to optimize US investor relations and corporate relations, and Ana Limón as Senior VP of Clinical Development and Global Medical Affairs to coordinate the US clinical activities and portfolio deployment. We finished this fourth quarter with a solid cash position of $32.5 million, which provides funding for further development of our exciting pipeline until Q1 2023.”
Fourth Quarter and Recent Highlights
Iadademstat in oncology:
Vafidemstat in large multifactorial CNS indications:
Vafidemstat in monogenic CNS indications:
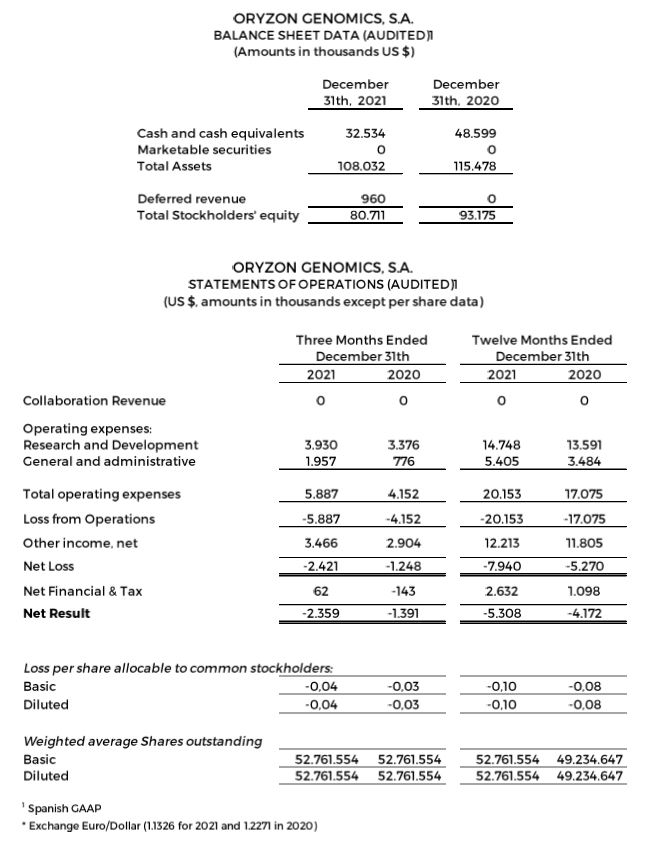
Financial Update: Fourth Quarter 2021 Financial Results
Research and development (R&D) expenses were $3.9 and $14.8 million for the quarter and 12 months ended December 31, 2021, compared to $3.4 and $13.6 million for the quarter and 12 months ended December 31, 2020.
General and administrative expenses were $2.0 and $5.4 million for the quarter and 12 months ended December 31, 2021, compared to $0.8 and $3.5 million for the quarter and 12 months ended December 31, 2020.
Net losses were $2.4 and $7.9 million for the quarter and 12 months ended December 31, 2021, compared to net losses of $1.2 and $5.3 million for the quarter and 12 months ended December 31, 2020. This is due to a higher investment in research and non-capitalized development of the ESCAPE clinical trial and non-recurring expenses, including $1.5 million in legal and regulatory preparations to adequate the company for a future Nasdaq dual listing. The result is in accordance with the specificity of the biotechnology business model, in the development phase of the Company, with a long-term maturation period for its products, and without recurrent income.
Negative net result of $5.3 million (-$0.10 per share) for the 12 months ended December 31, 2021, compared to a negative net result of $4.2 million (- $0,08 per share) for the 12 months ended December 31, 2020.
Cash, cash equivalents and marketable securities totaled $32.5 million as of December 31, 2021, compared to $48.6 million as of December 31, 2020.
During the year, Oryzon has obtained a grant of $1 million to support a new clinical trial (the HOPE trial) in patients with Kabuki syndrome.
Founded in 2000 in Barcelona, Spain, Oryzon (ISIN Code: ES0167733015) is a clinical stage biopharmaceutical company considered as the European champion in Epigenetics. Oryzon has one of the strongest portfolios in the field. Oryzon’s LSD1 program has rendered two compounds, vafidemstat and iadademstat, in Phase II clinical trials. In addition, Oryzon has ongoing programs for developing inhibitors against other epigenetic targets. Oryzon has a strong technological platform for biomarker identification and performs biomarker and target validation for a variety of malignant and neurological diseases. Oryzon has offices in Spain and the United States. Oryzon is one of the most liquid biotech stocks in Europe with +90 M shares negotiated in 2020 (ORY:SM / ORY.MC / ORYZF US OTC mkt). For more information, visit www.oryzon.com
Iadademstat (ORY-1001) is a small oral molecule, which acts as a highly selective inhibitor of the epigenetic enzyme LSD1 and has a powerful differentiating effect in hematologic cancers (see Maes et al., Cancer Cell 2018 Mar 12; 33 (3): 495-511.e12.doi: 10.1016 / j.ccell.2018.02.002.). A FiM Phase I/IIa clinical trial with iadademstat in R/R AML patients demonstrated the safety and good tolerability of the drug and preliminary signs of antileukemic activity, including a CRi (see Salamero et al, J Clin Oncol, 2020, 38(36): 4260-4273. doi: 10.1200/JCO.19.03250). In a still ongoing Phase IIa trial in elder 1L-AML patients (ALICE trial), iadademstat has shown encouraging safety and efficacy data in combination with azacitidine. Beyond hematological cancers, the inhibition of LSD1 has been proposed as a valid therapeutic approach in some solid tumors such as small cell lung cancer (SCLC), neuroendocrine tumors, medulloblastoma and others. In a Phase IIa trial in combination with platinum/etoposide in second line ED-SCLC patients (CLEPSIDRA trial, finalized), preliminary efficacy results have been reported. In total iadademstat has been tested in four clinical trials in more than 100 patients.
Vafidemstat (ORY-2001) is an oral, CNS optimized LSD1 inhibitor. The molecule acts on several levels: it reduces cognitive impairment, including memory loss and neuroinflammation, and at the same time has neuroprotective effects. In animal studies vafidemstat not only restores memory but reduces the exacerbated aggressiveness of SAMP8 mice, a model for accelerated aging and Alzheimer’s disease (AD), to normal levels and also reduces social avoidance and enhances sociability in murine models. In addition, vafidemstat exhibits fast, strong and durable efficacy in several preclinical models of multiple sclerosis (MS). Oryzon has performed two Phase IIa clinical trials in aggressiveness in patients with different psychiatric disorders (REIMAGINE) and in aggressive/agitated patients with moderate or severe AD (REIMAGINE-AD), with positive clinical results reported in both. Additional finalized Phase IIa clinical trials with vafidemstat include the ETHERAL trial in patients with Mild to Moderate AD, where a significant reduction of the inflammatory biomarker YKL40 has been observed after 6 and 12 months of treatment, and the pilot, small scale SATEEN trial in Relapse-Remitting and Secondary Progressive MS, where antiinflammatory activity has also been observed. Vafidemstat has also been tested in a Phase II in severe Covid-19 patients (ESCAPE) assessing the capability of the drug to prevent ARDS, one of the most severe complications of the viral infection, where it showed significant anti-inflammatory effects in severe Covid-19 patients. Currently, vafidemstat is in two Phase IIb trials in borderline personality disorder (PORTICO) and in schizophrenia patients (EVOLUTION). The company is also deploying a CNS precision medicine approach with vafidemstat in genetically-defined patient subpopulations of certain CNS disorders.