Do you want to obtain the Plasma Master File (PMF) certification?
For the marketing authorisation of plasma-derived medicinal products, the plasma master file (PMF) certification is used. The PMF is a compilation of all the required scientific data on the quality, safety and efficacy of human plasma relevant to the medicines, medical devices and investigational products. These data cover all aspects of the use of plasma, from collection to plasma pool.
The PMF is a separate set of documentation from the dossier for a medicine's marketing authorisation, and all applications should be submitted electronically in Electronic Common Technical Document (eCTD) format.
At Asphalion, we would be happy to support you with the PMF preparation and submission to the EMA.
Contact us if you need any help at info@asphalion.com


El equipo de investigadores observó cambios en el...

El gen AtCDF3 promueve una mayor producción de az...

Un estudio con datos de los últimos 35 años, ind...

En nuestro post hablamos sobre este interesante tipo de célula del si...
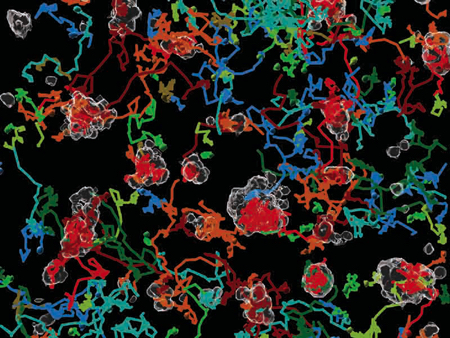
La revista ‘Nature Protocols’ selecciona esta técnica como “pro...
Portal de biotecnología en España
¡Suscríbase a nuestro newsletter para estar al día con las últimas noticias y ofertas!
2013 © Biotech-Spain.com - Site Developments SL. Todos los derechos reservados. Terminos y Condiciones | Política de Privacidad
Articles
Directory
