The European Medicines Agency (EMA) released a draft guideline on the quality requirements for medical devices (MD) in human medicines that include a MD (known as drug device combinations or DDC) for a 3-month public consultation, as part of a series related to the European’s medical devices regulation (MDR). The draft guideline covers devices that are necessary for the administration, dosing or use of the medicine. They can be devices that are integral to the medicinal product (integral DDC), or co-packaged with the medicinal product or referenced in the medicinal product information but obtained separately (non-integral DDC). It specifies which information about the device needs to be submitted as part of the initial marketing authorisation application (MAA) and subsequently during the product lifecycle.
A new template for the notified body (NB) opinion on the conformity of a device to the relevant general safety and performance requirements (GSPRs) laid down in MDR is proposed in the draft guideline.
The regulators will consider comments received during the public consultation (until 31 August 2019) with the goal to finalising the guideline before the regulation fully applies on 26 May 2020.

The research team observed changes in head circumf...

AtCDF3 gene induced greater production of sugars a...

Un estudio con datos de los últimos 35 años, ind...

En nuestro post hablamos sobre este interesante tipo de célula del...
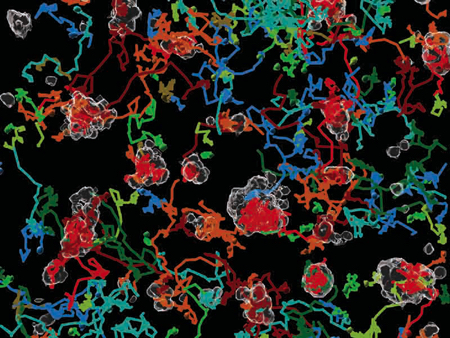
La revista ‘Nature Protocols’ selecciona esta técnica como “pro...
Biotechnology portal in Spain
Subscribe to our newsletter and stay up to date with the latest news and deals!
2013 © Biotech-Spain.com - Site Developments SL. All Rights Reserved. Terms of Service | Privacy Policy
Articles
Directory