After an acute heart lesion, such as a myocardial infarction, the human heart is unable to regenerate. The adult cardiac cells cannot grow and divide to replace the damaged ones, and the lesion becomes irreversible. But this does not happen in all animals. A freshwater fish native to Southeast Asia, known as a zebrafish, can completely regenerate its heart even after 20% ventricular amputation.
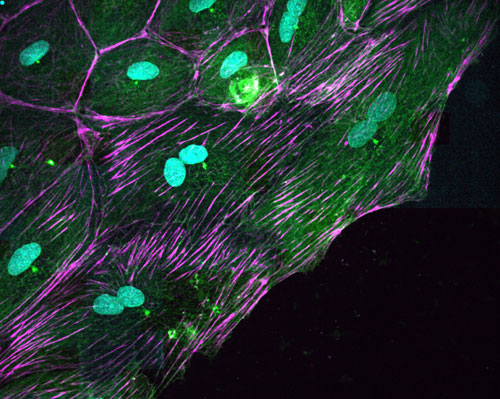
Cells of the epicardium of the zebrafish with two nuclei (in blue)
This extraordinary regenerative capacity has attracted the attention of researchers from all over the world, who see the range of possibilities that would be opened up if this mechanism of cell regeneration could be applied in human therapies.
In an article published today in the Nature Materials journal, a team of researchers from the Institute of Bioengineering of Catalonia (IBEC) led by Xavier Trepat, in collaboration with the Centre for Regenerative Medicine in Barcelona (CMR [B]), have discovered a surprising mechanism by which zebrafish heart cells move and divide during regeneration.
Researchers have focused on the epicardium, which is the layer of cells on the outer surface of the heart. Although the epicardium cells represent only a small fraction of the heart’s mass, they play a fundamental role in its regeneration. “The epicardium is the origin of several of the heart’s cell types, and secretes biochemical signals that tell the cells what they have to do at all times. It’s a kind of regeneration ‘hub’”, states Angel Raya, ICREA Researcher and director of CMRB.
After a heart lesion, the epicardium cells begin to divide and move en masse to cover the wound. Researchers have observed that, during this process, the cells become binucleated: they duplicate the genetic material and separate it into two nuclei, but they are not divided into two independent cells. “We were very surprised to discover cells that, instead of having one nucleus, as is the case in most tissues, they have two nuclei, and each of them contains a copy of the cell’s DNA” says Trepat, ICREA researcher at IBEC and associate professor of the University of Barcelona.
Researchers have discovered that the mechanism by which cells become binucleated has a biomechanical origin. Once DNA has already separated into two nuclei, most animal cells form a contractile ring at its centre. As it contracts, this ring divides the mother cell into two daughter cells. In the case of the heart cells of the zebrafish, the study shows that the ring adheres to the fibres of its environment so that it cannot tighten. The result is that the two daughter cells cannot separate despite having correctly duplicated their DNA.
“Multinucleation is a well-known phenomenon in cancer, because it is a cause of genetic instability. In other words, cancer cells lose control of the proteins they synthesise and behave pathologically. In the case of the heart of zebrafish, the multinucleation is physiological and does not seem to cause any problem”, states Marina Uroz, the article’s main author. The next step will be to study the role of multinucleated cells during the regeneration of the heart and other organs.
Dr. Trepat and Dr. Raya are part of CIBER-BBN (Biomedical Research Networking Centre in Bioengineering, Biomaterials and Nanomedicine)
Reference article: M. Uroz, A. Garcia-Puig, I. Tekeli, A. Elosegui-Artola, J. F. Abenza, A. Marín-Llauradó, S. Pujals, V. Conte, L. Albertazzi, P. Roca-Cusachs, A. Raya and X. Trepat. Traction forces at the cytokinetic ring regulate cell division and polyploidy in the migrating zebrafish epicardium. Nature Materials, DOI: 10.1038/s41563-019-0381-9(2019)