During the day of March 21 from 10:00 a.m. to 4:00 p.m., Konexio Biotech had the honor of being present at the 'FIRST DAY OF INSPECTION AND CONTROL OF MEDICINES AEMPS & AEFI' (Spanish Agency for Medicines and Health Products and Spanish Association of Pharmaceutical Industry), which was in the main Casa de Burgos of the provinces, Madrid.
As the AEFI itself points out, regulations evolve, their application does so with them and knowing them is vital for their implementation in the Industry. With the help of experts from the AEMPS and in collaboration with AEFI and industry experts, in these first days of pharmaceutical inspection we will discover how to do it.
Our colleague and CEO, Andrea Romero López was part of the last table, together with María Gemma Fernández Miguel (NCF Inspector, AEMPS) and moderated by Marta Rodríguez Vélez (Voice of Quality Assurance, Manufacturing and Quality Control. AEFI, Section Center).
In it, they discussed near-patient manufacturing in the context of Advanced Therapies, Part IV of Good Manufacturing Standards (GMP) and decentralized manufacturing.
From Konexio Biotech, we would like to greatly thank AEFI for this opportunity, counting on us and making us participate in this essential event in the sector, whose complete information is available at: https://aefi.org/eventos/primera-jornada-de -inspection-and-control-of-medicines-aemps-aefi/.
'Connect to Advance. Be Biotech'

The research team observed changes in head circumf...

AtCDF3 gene induced greater production of sugars a...

Un estudio con datos de los últimos 35 años, ind...

En nuestro post hablamos sobre este interesante tipo de célula del...
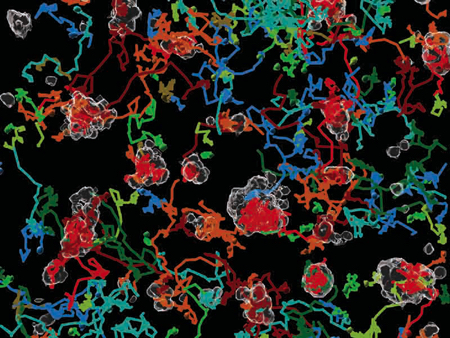
La revista ‘Nature Protocols’ selecciona esta técnica como “pro...
Biotechnology portal in Spain
Subscribe to our newsletter and stay up to date with the latest news and deals!
2013 © Biotech-Spain.com - Site Developments SL. All Rights Reserved. Terms of Service | Privacy Policy
Articles
Directory
